Sustainability
Responsible producer and distributor
AddLife's role in the value chain, mainly as a distributor, implies to both have a close dialogue with many customers on local level as well as to chose and influence among global leading suppliers. This is a unique opportunity to support a sustainable transition with our partners and together develop solutions that could also have a positive business impact.
 The 2021 materiality assessment combined with the stakeholder dialogues shows that product quality and service, responsible sourcing and business model innovation are prioritised areas for Addlife for future growth. We will continue working with the material topics during 2022. For example, we will identify most relevant focus areas, integrate these into strategy and create concrete actions and KPIs.
The 2021 materiality assessment combined with the stakeholder dialogues shows that product quality and service, responsible sourcing and business model innovation are prioritised areas for Addlife for future growth. We will continue working with the material topics during 2022. For example, we will identify most relevant focus areas, integrate these into strategy and create concrete actions and KPIs.
In parallel with the materiality assessment, AddLife has evaluated how to strengthen the measurement of sustainability data, to increase transparency and enable future comparison. Implementation to be completed during 2022.
The majority of AddLife’s product range is produced by leading suppliers around the world. As a distributor, we mainly have an indirect impact on matters such as choice of materials, raw materials, packaging and terms of employment. None of the Group’s Swedish subsidiaries engage in activities that require a permit or notification under the Swedish Environmental Code. None of the foreign subsidiaries engage in activities subject to equivalent requirements for notification or permits. None of the Group’s companies are engaged in any environment-related disputes.
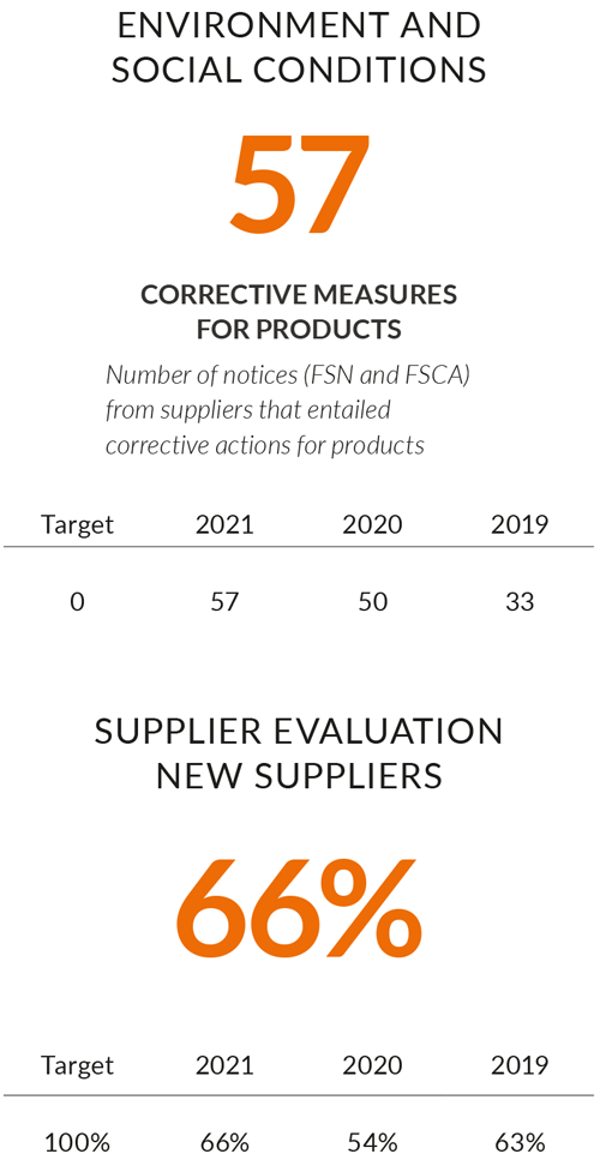
AddLife aims to work closely with suppliers to achieve long-term and sustainable development. The goal is for all suppliers to live up to AddLife’s code of conduct and environmental requirements.
Furthermore, AddLife encourages suppliers to respect fundamental human rights and to treat employees in accordance with the ILO declaration on fundamental principles and rights at work.
Products must be CE-certified before they can be marketed in Europe. The new EU directive for medical devices, MDR, was implemented in 2021 and the new EU directive for in vitro diagnostics products, IVDR, has been delayed. For some products, these new directives may require renewed testing for CE-certification, which may be both expensive and challenging for small operators with limited resources and cause larger global players to opt out of smaller local markets.
During the year, AddLife has managed the MDR implementation well, without loosing any supplier due to non-compliance. AddLife is working proactively through industry organisations to stay up to date regarding the IVDR implementation, though each company in the group is responsible for handling their suppliers and processes. Our subsidiaries continually work to assess new and existing suppliers. During 2022, AddLife will implement a new sustainability management system, which will be mandatory for all subsidiaries. This will allow an improved monitoring of the group's sustainability performance. AddLife also aims to increase the sustainability requirements for our suppliers across all companies in the group and improve the assessment of material or alternatives to material of our own manufactured products.
The market situation during 2021 has still been complicated due to the pandemic. AddLife’s has been one of the key players to successfully supply the health systems with much needed diagnostic tests and other critical products, despite challenges in logistics, supply chain etc.
Due to strategic acquisitions, with large implications on the group's market presence, the foreseen geographic mapping of supply chain has not been completed.
- Developed the process for evaluation and approval of new suppliers
- Introduction of green car policy
- Evaluation of sustainability management system
- Implementation of common supplier sustainability evaluation
- Implementation of green car policy
- Implementation of sustainability management system